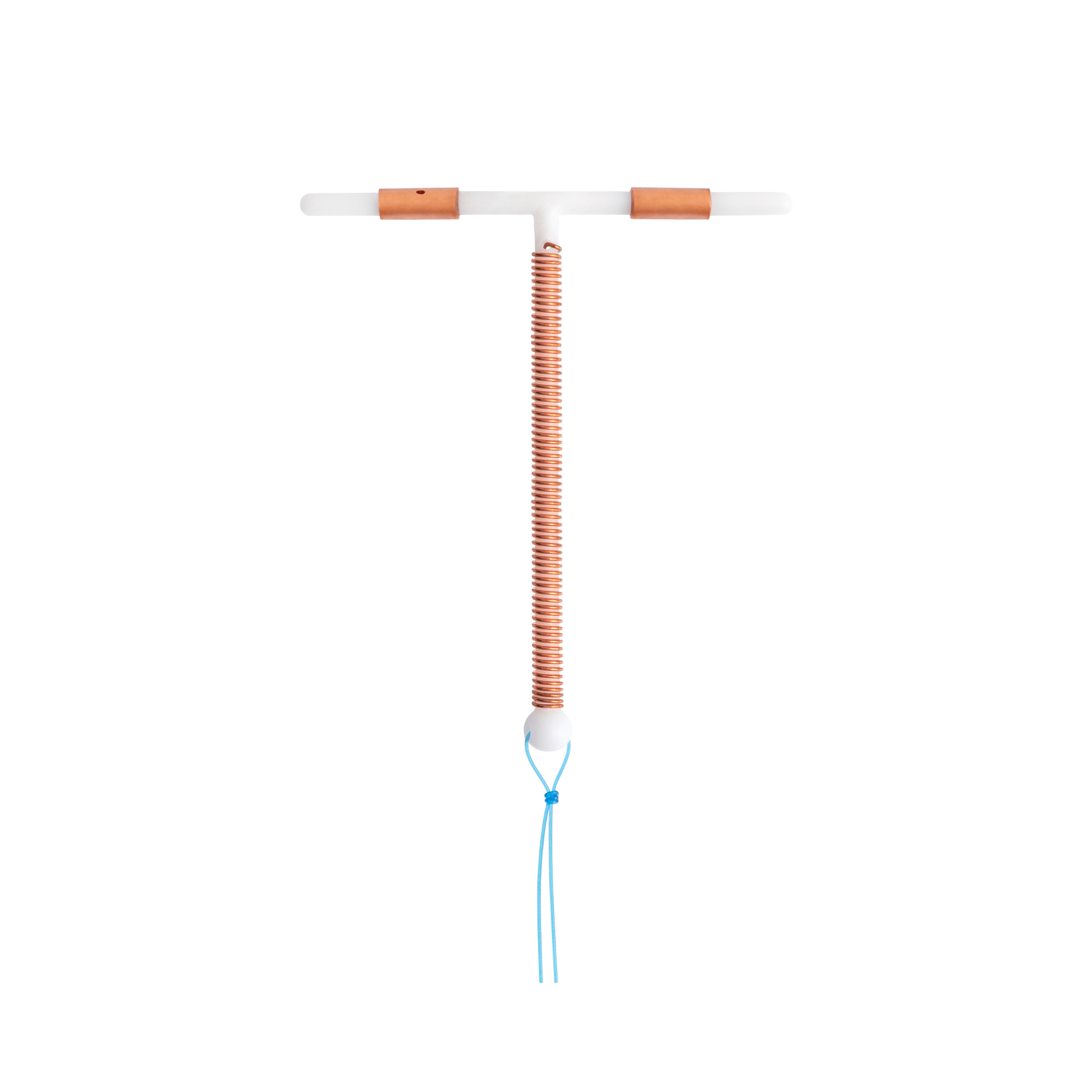
Etherena® T Cu 380A
Brand: Etherena®Intrauterine contraceptive device (IUD) with 1-step insertion system
380 mm² copper intrauterine contraceptive device (IUD) with insertion system and disposable hysterometer.
The T-IUD is non-hormonal, it prevents implantation in the uterus of the fertilized oocyte. This activity is reinforced in an ancillary manner by the presence of copper. Copper has a cytotoxic effect on gametes which causes damage to sperm, and inhibits fertilization. The Etherena® T-IUD is effective from the day it has been inserted.